Research Funding
Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA
Background
KEYNOTE-199 (NCT02787005) is a multicohort phase 2 study. Cohort (C)4 (RECIST-measurable disease) and C5 (bone-predominant disease) consist of chemotherapy-naive patients (pts) with mCRPC treated with enza + pembro after progression with enza. Results for C4 and C5 presented.
Methods
Pts with or without prior abiraterone had clinically meaningful response/benefit to enza followed by disease progression. Pts received pembro 200 mg Q3W with continuation of enza for up to 35 cycles or until progression/intolerable toxicity. Primary end point: ORR, blinded independent central review (C4). Secondary end points: DCR, PSA response rate (≥50% reduction), rPFS, OS, and safety (C4, C5); DOR (C4).
Results
Of 126 pts (C4, 81; C5, 45), 107 discontinued, primarily due to progression. Median follow-up: 13.7 mo (C4, 11.8; C5, 18.6). ORR (95% CI) for pts with measurable disease was 12% (6-22) in C4; DCR for all pts: 51% (39-62) in C4 and 51% (36-66) in C5 (Table). Any grade/grade 3-5 treatment-related AEs occurred in 75%/26% pts in C4 and 69%/24% in C5. Two pts in C4 died of immune-related AEs (Miller Fisher syndrome and myasthenia gravis). Incidence of any grade/grade 3-4 rash (regardless of treatment relatedness) was higher than previously reported for individual agents (33%/6%). All except one pt (grade 3 treated with IV steroids) were treated with oral/topical steroids or had no intervention.
Conclusions
Addition of pembro to enza following enza resistance showed modest antitumor activity in pts with RECIST-measurable and bone-predominant mCRPC. Combination had manageable safety and is being evaluated in a phase 3 trial. Clinical trial information: NCT02787005
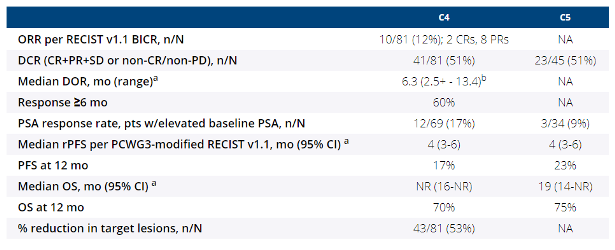
a Product-limit (Kaplan-Meier) method for censored data
b ‘+’ indicates ongoing responder